

A 37-year-old male from California presented to the Tropical Disease Clinic with a suspect malaria infection. He had returned from approximately 10 months in various areas in Africa, including Uganda and Kenya. During his trip he used an untreated bed net and was taking appropriate prophylaxis. However, he did indicate he skipped several doses while in Uganda and Kenya. On his original return, he had been screened for malaria using routine microscopy and PCR for Plasmodium vivax and P. falciparum, all of which were negative. Prior to being seen in the Tropical Disease Clinic, he had been seen elsewhere and a P. vivax infection had been diagnosed on the basis of microscopy. He had been treated appropriately for P. vivax. He improved, but three weeks later he presented with fever and fatigue; at this point he was seen at the Tropical Disease Clinic. The following images were seen; this same picture was diagnosed as P. vivax prior to visiting the Tropical Medicine Clinic. Because there were some questions raised regarding the species identification, more extensive molecular testing was performed.
![]()
![]()
![]()
![]()
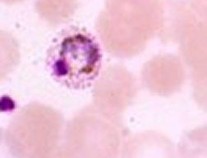
What organisms are suggested from the images shown above?
How does his history impact possible parasitic infections?
Does the morphology provide an accurate identification? Why or why not? What might this parasite mimic?
What are the pros and cons of routine microscopy, rapid testing, and molecular testing for the diagnosis of Plasmodium to the species level?
The images certainly indicate an infection with Plasmodium species; however, it is easy to see how these organisms might be confused with P. vivax rather than P. ovale.
The patient history is consistent with several species of Plasmodium; since
P. vivax and P. falciparum are the most common, it was practical to screen for these two species.
The overall morphology of P. vivax and P. ovale can be very similar; not all stages can be clearly assigned to one or the other species. More characteristic organisms belonging to the two species can be seen below.
As seen from this case, the use of routine microscopy can be problematic in identifying the organisms as either P. vivax or P. ovale. The use of malaria rapid cartridge tests does not identify P. ovale as accurately as P. vivax or P. falciparum. Consequently, the use of molecular methods provides a much more accurate identification to species for the five species of human malaria (rapid test methods are under development for Plasmodium knowlesi).
COMMENTS ON THE PATIENT and INFECTION:
This case highlights the need to consider P. ovale as a cause of imported malaria and to include its detection, as well as P. malariae, in imported malaria screening programs. This patient presented particular diagnostic challenges that resulted in a failure of early detection and diagnosis of P. ovale infection based on the microscopic examination of the blood films. Distinguishing P. ovale from P. vivax by microscopy, already difficult due to the species’ morphologic similarity, can lead to inaccurate species identifications.
COMMENTS ON THE METHOD RECOMMENDATIONS:
The routine microscopic examination of stained blood films can be difficult, particularly in the accurate identification to the species level; P. vivax and P. ovale tend to resemble one another and can easily be confused.
Differentiation to the species level can be a particular problem for all five species of human malaria when ring forms only are seen in the blood films.
COMMENTS ON THE IMAGES:
The images seen in the original smears are difficult to differentiate between P. vivax and P. ovale. More characteristic images of all five species of human malaria can be seen below. Row 1 is P. vivax; row 2 is P. ovale; row 3 is P. malariae; row 4 is P. falciparum; row 5 is P. knowlesi.
Garcia, L.S. 2016. Diagnostic Medical Parasitology, 6th Ed., ASM Press, Washington, D.C.
1 2 3 4 5
|
|
|
|
| |||||
|
|
|
|
| |||||
|
|
|
|
| |||||
|
|
|
|
| |||||
|
|
|
|
| |||||
|
|
|
|
|
48-hour cycle
Tends to infect young cells
Enlarged RBCs
Schüffner's dots (true stippling) after 8-10 hours
Delicate ring
Very ameboid trophozoite
Mature schizont contains 12-24 merozoites
72-hour cycle (long incubation period)
Tends to infect old cells
Normal size RBCs
No stippling
Thick ring, large nucleus
Trophozoite tends to form "bands" across the cell
Mature schizont contains 6-12 merozoites
48-hour cycle
Tends to infect young cells
Enlarged RBCs with fimbriated edges (oval); usually one end of RBC only – different from crenated RBCs
Schüffner's dots appear in the beginning (in RBCs with very young ring forms in contrast to P. vivax)
Smaller ring than P. vivax
Trophozoite less ameboid than that of P. vivax
Mature schizont contains average 8 merozoites
36-48-hour cycle
Tends to infect any cell regardless of age, thus very heavy infection may result
All sizes of RBCs
No Schüffner's dots (Maurer's dots: may be larger, single dots, bluish)
Multiple rings/cell (only young rings, gametocytes, and occasional mature schizonts are seen in peripheral blood)
Delicate rings, may have two dots of chromatin/ring, appliqué or accolé forms
Crescent-shaped gametocytes
24-hour cycle
Tends to infect any cell regardless of age, thus very heavy infection may result
All sizes of RBCs, but most tend to be normal size
No Schüffner’s dots (faint, clumpy dots later in cycle)
Multiple rings/cell (may have 2-3)
Delicate rings, may have two or three dots of chromatin/ring, appliqué forms
Band form trophozoites commonly seen
Mature schizont contains 16 merozoites, no rosettes
Gametocytes round, tend to fill the cell
*Early stages mimic P. falciparum; later stages mimic P. malariae
Each Quiz has a two section format: the first section will present the Quiz topic and the second section will provide a discussion of the answer and/or various options in response to the Quiz situation presented to the user. In some situations, there may be more than one correct response.
The content within this site is made possible through the extensive contribution of Lynne S. Garcia, M.S., MT(ASCP), CLS(NCA), BLM(AAB), F(AAM), Director, Consultantation and Training Services (Diagnostic Medical Parasitology and Health Care Administration). For additional information, she can be contacted at LynneGarcia2@verizon.net.
Reference: Garcia, L.S. 2015. Diagnostic Medical Parasitology, 6th Ed., ASM Press, Washington, D.C.